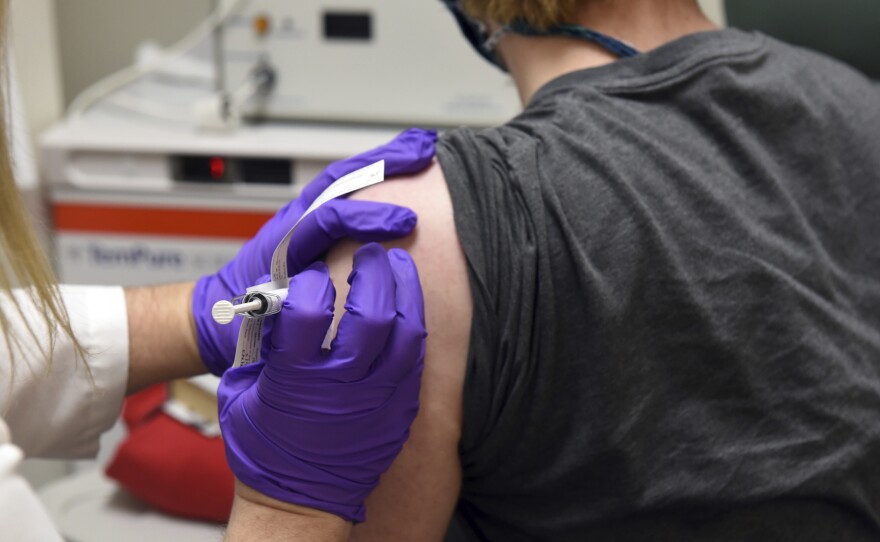
It takes a lot of paperwork to be a human test subject. COVID-19 vaccine trial volunteer Christian Ramers filled out multiple forms before his pen ran dry.
He agreed to allowing his blood to be used for further COVID-19 study, authorized sharing of his health information and answered confidential questions from a clinician that also had her scribbling down answers.
Joining a COVID-19 vaccine study is lengthy and invasive and the personal gain is uncertain. While participants may enroll because of altruism, trials are completely voluntary and double blind to protect the study’s integrity — neither participants nor researchers know who receives the experimental vaccine over a placebo.
Experts are concerned that volunteers will drop out of ongoing trials if the FDA grants emergency use approval to a vaccine, which could be later this month. They are worried participants may instead seek the approved immunization and leave researchers without crucial follow-up data required to produce multiple vaccines the country needs.
Ramers, a community clinic physician and member of San Diego County’s clinical vaccine advisory group, said an emergency approved tool is urgently needed, but continuing other studies is also crucial.

“We're in a situation where we want to get something approved as quickly as possible, and yet we also need all the information we can get from these,” he said while sitting in the clinic where he received an injection for the Janssen Pharmaceuticals trial.
Ramers said he joined the trial to fight against the virus that’s sickening his patients at Family Health Centers of San Diego, but also to combat vaccine skepticism among communities of color.
“There's a lot of suspicion and fear, and that's based in historical injustices, really, from biomedical research, and so I wanted to really take a proactive stance against that,” said Ramers, who is Cuban American.
But he left the door open for seeking that first emergency approved vaccine — a CDC advisory panel on Tuesday said health care workers should be among the first to receive it.
“If I have the ability to get a vaccine that's approved and I get in line because I'm a health care provider, you always have the ability to pull out of the vaccine trial,” Ramers said.
Limited access
UC San Diego Health’s Susan Little, the principal investigator on the local trial for Janssen, which is owned by Johnson & Johnson, said volunteer dropoff is a real concern. She said she’s already planning on how to speak with participants who are considering dropping out — the first emergency use approval could be granted to Pfizer after an FDA advisory committee meeting on Dec. 10.
“We might advise our participants that, really if you were to wait until February, you could evaluate this vaccine study that you're on and see if it was better,” said Little, who is also overseeing the local AstraZeneca trial.
She stressed participants can withdraw at any point without reason nor penalty, but worried many may not realize early doses will be in short supply even among identified priority groups.
“There’s likely to be a very limited number of doses available for the entire country,” Little said.
Gov. Gavin Newsom said this week California expects to receive 327,000 doses from Pfizer in mid-December.
As a doctor, Ramers could be among the few who receive it. He said he hasn’t made up his mind about seeking it, but will assess when the detailed data is released.
“I'll cross that bridge when I come to it,” he said.
Research after injection

Early research from leading vaccine contenders Pfizer, Moderna and AstraZeneca showed high levels of efficacy, but a serendipitous dosing error by AstraZeneca did raise questions. Still, the companies reported minimal safety concerns that were on par with what’s seen after a flu shot.
But UCSD Health’s Davey Smith, a lead on COVID-19 treatment trials, said those are short-term findings. Applications for emergency use should include about two months of follow-up data, according to FDA guidelines published in October.
Adverse effects would likely show up in that window, but Smith said long-term efficacy and safety monitoring is needed before officials grant full license to use the vaccine on the general public.
“It doesn’t mean that we have any preconceived notion that these are going to be harmful long term, I’m not saying that at all, it's just that the FDA likes to see longer-term data,” Smith said.
That’s complicated if volunteers drop out after the first emergency approved vaccine. But their continued participation, especially those unknowingly in the placebo cohort, presents ethical dilemmas.
Unblinding participants
Arthur Caplan, a bioethics professor at the NYU Grossman School of Medicine said participants may demand to know whether or not they’re receiving the experimental vaccine and instead received a dummy shot, and pharmaceutical companies should comply.
“They're bravely volunteering, but at some point, if we learn new information that we get a vaccine that works, you’ve got to tell them,” Caplan said.
Full Janssen statement to KPBS
“We are incredibly grateful for the contribution of those who have volunteered to participate in our clinical trials and in all the COVID vaccine clinical trials.
“Participants in Janssen’s Phase 3 COVID-19 vaccine study will not be disadvantaged with regard to receiving a vaccine that has been approved under Emergency Use Authorization. While we anticipate that vaccines will be made available in the near future, healthcare workers and at-risk populations will be prioritized and the vaccination of broad populations will take time.
“It is very important that multiple COVID-19 vaccine approaches are pursued to find the most effective vaccines and to help meet the world demand for doses.
“We plan on having transparent communication to clinical trial participants, as described in the consent form of the study, regarding new information which may affect their participation in the study. Therefore, when vaccine options become available, information will be shared on their availability (including which populations are able to receive the vaccine) in a way that permits an individual to make an autonomous, informed choice about whether to continue blinded participation in the trial or seek to be unblinded, to potentially access a newly approved vaccine (and depending on whether they were assigned to the active vaccine or the placebo).”
But he said the solution isn’t to give all placebo recipients the real injection.
“Some people would say give it to everybody in the group because they deserve it having volunteered, but if they're not high-risk, they don't need it yet,” Caplan said.
According to the FDA guidelines, the agency “does not consider availability of a COVID-19 vaccine under EUA, in and of itself, as grounds for stopping blinded follow-up in an ongoing clinical trial.”
Applicants have to include how they’ll ensure long-term study, the document said.
A Janssen spokesperson told KPBS the company will notify participants about eligibility criteria for an emergency approved vaccine.
In an emailed statement, the spokesperson said information will be shared “in a way that permits an individual to make an autonomous, informed choice about whether to continue blinded participation in the trial or seek to be unblinded, to potentially access a newly approved vaccine."
Pfizer shared with KPBS a letter it sent to participants that said the company was “exploring potential ways to change the study to create a process that would allow interested participants in the placebo group who meet the eligibility criteria for early access in their country to ‘cross-over’ to the vaccine group in the study.”
The Nov. 9 document said such a change would require regulatory approval.